–1899
Era I The Classical Crisis
By the late 19th century, classical physics — Newton's mechanics and Maxwell's electromagnetism — had achieved extraordinary successes. Yet two phenomena stubbornly resisted explanation: the spectrum of radiation emitted by hot objects (blackbody radiation) and the discrete lines of atomic spectra. These were not minor puzzles. They signalled that something was fundamentally wrong.
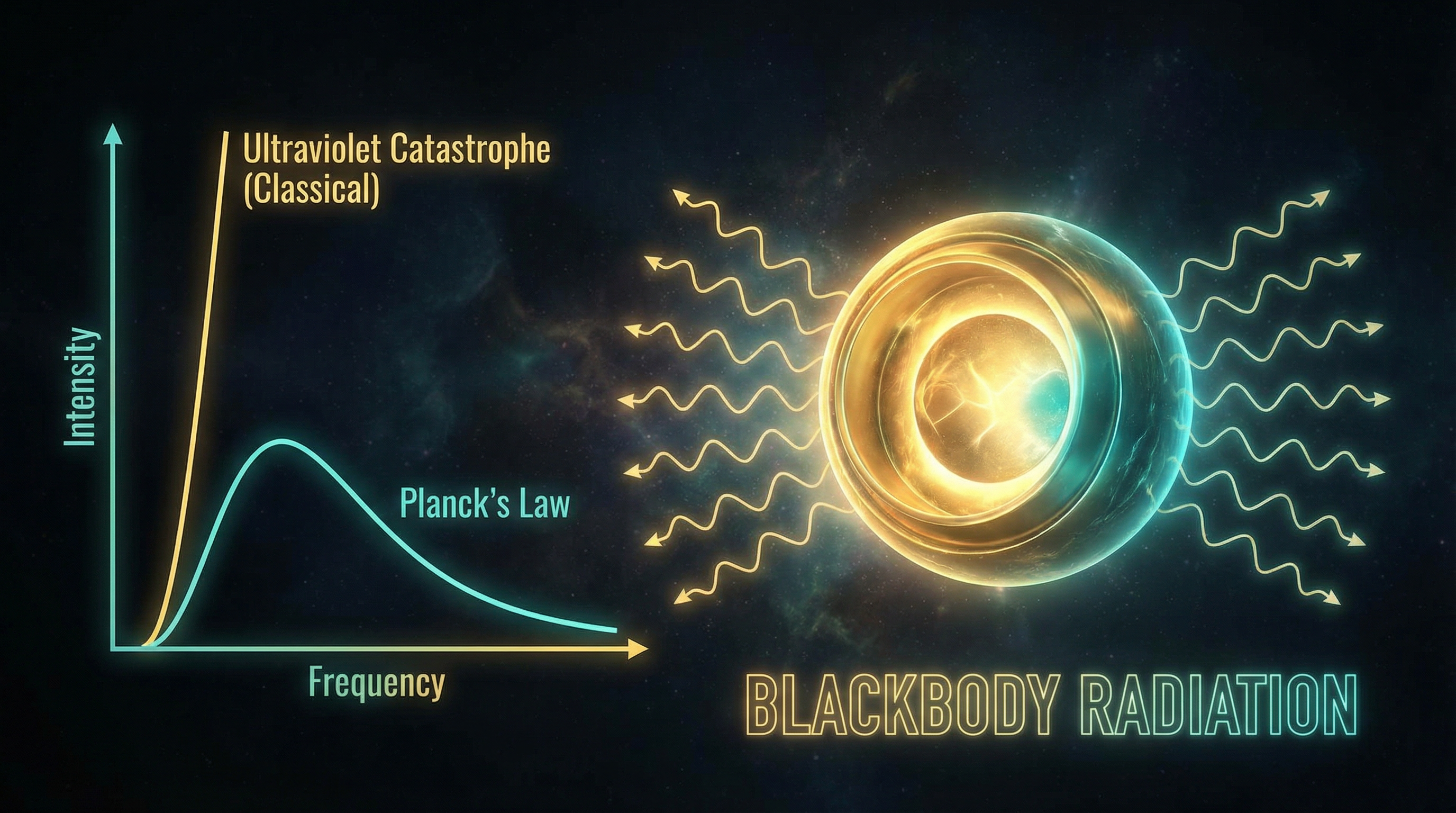
The Blackbody Problem
A perfect blackbody absorbs and re-emits all radiation. Classical thermodynamics predicts the Rayleigh–Jeans law for the power spectrum — which diverges at high frequencies (the "ultraviolet catastrophe"). No classical model could match the observed curve across all wavelengths.
Spectral Lines & Empirical Formulas
Johann Balmer (1885), Johannes Rydberg (1888), and Walter Ritz (1908) identified patterns in the wavelengths of hydrogen's emission lines. These elegant numerical relations had no theoretical explanation in classical physics — the atom was a black box.
Models of the Atom
J.J. Thomson (1897) discovered the electron. His "plum-cake" model placed electrons embedded in a diffuse sphere of positive charge. Ernest Rutherford's gold-foil experiments (Marsden and Geiger, 1908–1911) shattered this picture: almost all the atom's mass was concentrated in a tiny positive nucleus, with electrons orbiting at a distance. But this planetary model was classically unstable — electrons should spiral in and radiate.
Era II — December 14, 1900 Planck & the Birth of the Quantum
Planck's Quantum Hypothesis
On December 14, 1900 — the birth date of quantum mechanics — Max Planck presented his derivation of the blackbody radiation law to the German Physical Society. He found that he could fit the experimental spectrum perfectly by assuming that energy is not exchanged continuously between matter and radiation, but only in discrete packets — quanta — of magnitude $E = h\nu$, where $\nu$ is the frequency.
Planck himself was uncomfortable with this assumption, viewing it initially as a mathematical trick rather than a physical reality. He would spend years trying to reconcile it with classical physics — without success. The genie was out of the bottle.
Era III — Annus Mirabilis Einstein & the Photon
In 1905 — his miraculous year — Einstein published the paper that would eventually earn him the Nobel Prize. He took Planck's quantum of energy seriously: light itself, he proposed, is composed of discrete particles — photons — each carrying energy $E = h\nu$ and momentum $p = \hbar k$.
The Photoelectric Effect
Classical wave theory predicted that increasing the intensity of light should eventually eject electrons regardless of frequency. Experiment disagreed: no electrons were emitted below a threshold frequency, no matter how bright the light. Einstein explained this perfectly with photons: each electron must absorb one photon; if $h\nu$ is less than the work function, no ejection occurs. The energy of ejected electrons depends only on frequency, not intensity.
Wave–Particle Duality Begins
Einstein's photon was deeply unsettling: Maxwell's equations clearly described light as a wave — interference and diffraction were well established. Yet here was compelling evidence that light also behaved as particles. This paradox — wave–particle duality — would become one of the central mysteries of quantum theory.
Einstein's Specific Heat
Einstein also applied quantization to the vibrations of atoms in a solid, explaining the anomalous specific heat of solids at low temperature — another classical failure. This extended Planck's idea far beyond blackbody radiation and showed that quantization was a universal feature of nature.

Era IV — 1913 The Bohr Atom & Old Quantum Theory
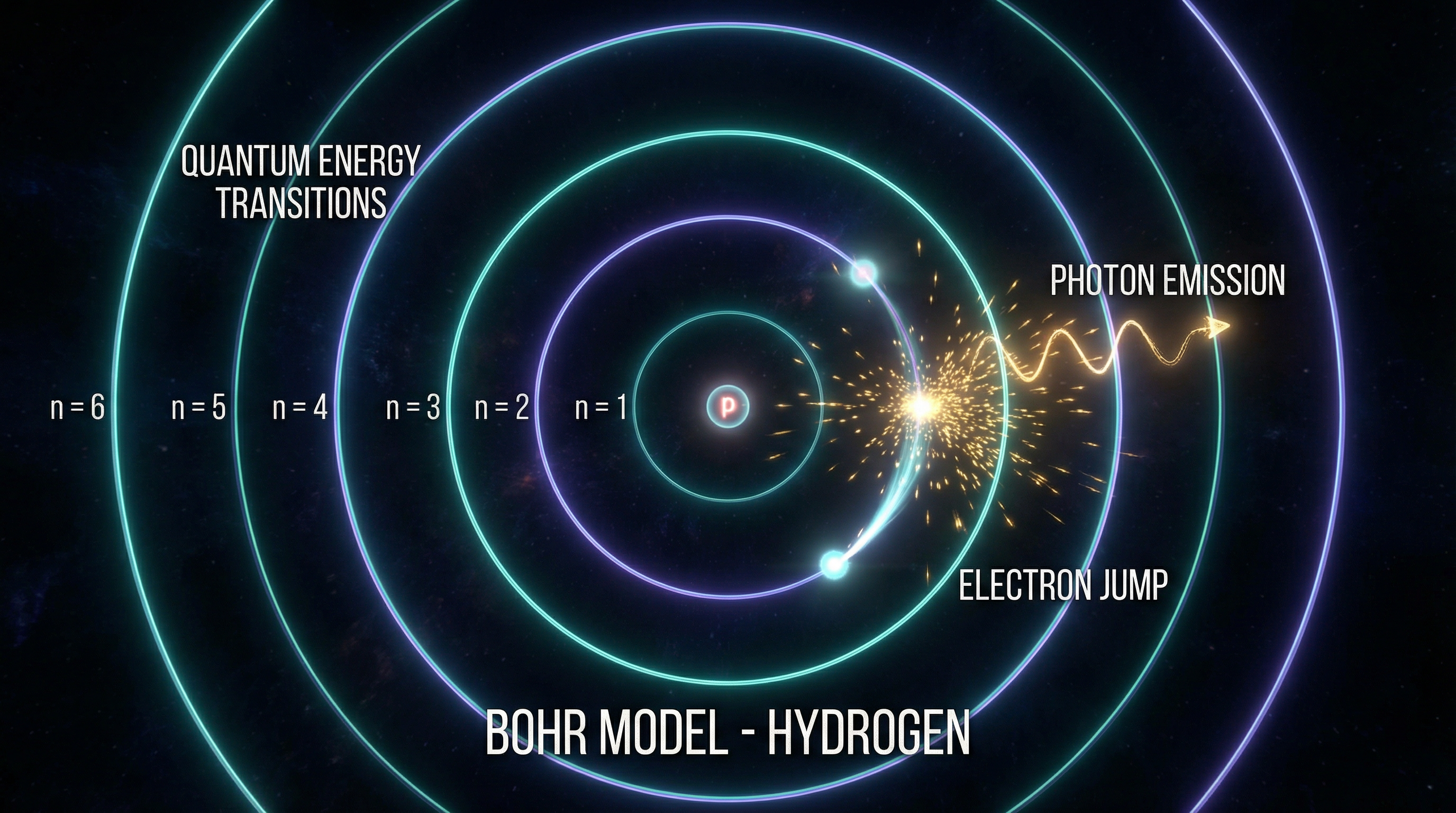
Bohr's Atomic Model
Niels Bohr combined Rutherford's nuclear atom with Planck's quantization rule. He postulated that electrons could only occupy certain discrete orbits — "stationary states" — in which they do not radiate. When an electron jumps between two levels, it emits or absorbs a photon whose frequency satisfies the Bohr frequency condition:
This was a complete break with classical mechanics: in classical electrodynamics, an accelerating electron always radiates. Bohr simply forbade this. The model correctly predicted the hydrogen spectrum — a stunning success — but had no deeper justification.
Angular Momentum Quantization
Paul Ehrenfest showed that the electron's angular momentum must be quantized: $L = n\hbar$. This "old quantum theory" rule, combined with Bohr's postulate, gave the correct energy levels. Bohr and Sommerfeld extended the model to elliptical orbits (1915–1916), introducing additional quantum numbers.
–1927
Era V Key Experiments
Quantum mechanics was not invented in a vacuum. A series of brilliant experiments forced theorists to abandon classical intuitions step by step.
Direct Evidence for Energy Quantization
James Franck and Gustav Hertz fired electrons through mercury vapor and measured the current as a function of accelerating voltage. They observed sharp dips every 4.9 V: electrons lose exactly 4.9 eV to mercury atoms, which then emit UV radiation at $\lambda = 253.7\ \text{nm}$. This was direct experimental proof that atomic energy levels are discrete — electrons can only transfer energy in exact quantum amounts. Franck and Hertz received the Nobel Prize in 1925.

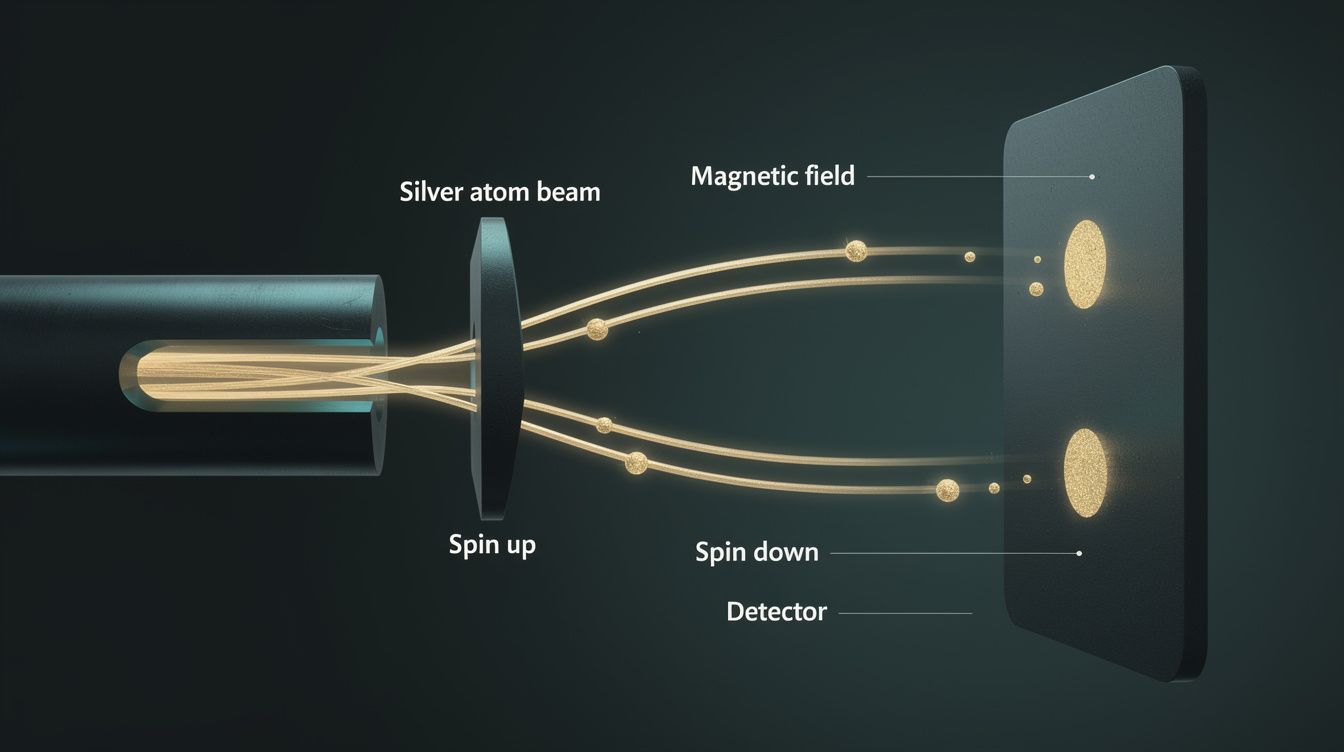
Space Quantization of Angular Momentum
Otto Stern and Walther Gerlach passed silver atoms through an inhomogeneous magnetic field and observed the beam split into two discrete spots on the detector plate — not a continuous smear as classical physics predicts. This demonstrated the quantization of angular momentum in space and gave the first indirect evidence for electron spin. The result was inexplicable until Pauli (1924) introduced a two-valued quantum number, and Uhlenbeck and Goudsmit (1925) proposed the electron has intrinsic spin $s = 1/2$.
Matter Has Wave Properties
Louis de Broglie proposed in his 1923 PhD thesis that, just as Einstein had shown light waves behave as particles (photons), particles of matter should also behave as waves. He assigned a wavelength to any particle with momentum $p$:
For a macroscopic object this wavelength is utterly negligible. For an electron it is on the order of atomic spacings — and therefore testable. Victor Henri, a physical chemist, learned of de Broglie's hypothesis and communicated it to Schrödinger in 1925, directly inspiring the wave equation.

Experimental Confirmation of Electron Diffraction
Clinton Davisson and Lester Germer at Bell Labs (1927) scattered electrons off a nickel crystal and observed interference maxima at exactly the angles predicted by de Broglie's formula — electron diffraction, the definitive proof of matter waves. Independently and almost simultaneously, George Paget Thomson (son of J.J. Thomson) demonstrated the same effect by passing electrons through thin metal foils. Both received the Nobel Prize in 1937.
The double irony: J.J. Thomson won the Nobel Prize for proving the electron is a particle; his son G.P. Thomson won it for proving the electron is a wave.
Wave–Particle Duality Made Vivid
In 1992, Shimizu and collaborators performed Young's double-slit experiment with individual neon atoms cooled to milliKelvin temperatures. Even single atoms, one at a time, built up an interference pattern on the detector. Each atom passes through both slits simultaneously — a direct, visceral demonstration that quantum superposition is a real phenomenon, not a theoretical abstraction.

Era VI — Summer 1925 Matrix Mechanics: Heisenberg, Born, Jordan & Dirac
Heisenberg's Breakthrough
Werner Heisenberg, on the island of Helgoland recovering from hay fever, had a radical insight: abandon the unobservable classical trajectory (position, momentum as functions of time) and build a mechanics based only on observable quantities — spectral line frequencies and intensities. He wrote down rules for multiplying arrays of transition amplitudes.
When Heisenberg showed his calculations to Max Born in Göttingen, Born recognised the arrays as matrices — a mathematical object he knew from linear algebra. The non-commutativity of matrix multiplication was the key: $XP \neq PX$.
Born–Heisenberg–Jordan Paper
Born, Heisenberg, and Jordan formalized the theory in a comprehensive paper establishing the rules of matrix mechanics. The commutation relation $[X,P] = i\hbar$ was central. In October 1925, Pauli solved the hydrogen atom in this framework — a spectacular validation.
Dirac at Cambridge
Paul Dirac, a 23-year-old student at Cambridge, received a copy of Heisenberg's paper in July 1925. Within weeks he had independently developed his own version of quantum mechanics, recognising the connection between the commutator $[X,P]$ and the classical Poisson bracket $\{x,p\} = 1$. Dirac's formulation would become the foundation of the modern theory.
Pauli's Exclusion Principle & Spin
Wolfgang Pauli (1924) introduced a fourth quantum number — a mysterious "two-valuedness" — to explain the fine structure of spectral lines and the periodic table. George Uhlenbeck and Samuel Goudsmit (1925) gave it a physical interpretation: the electron possesses intrinsic angular momentum, or spin, with $s = 1/2$. Pauli's exclusion principle — no two fermions can occupy the same quantum state — is the foundation of all of chemistry.

1926
Era VII — 1925–1926 Schrödinger & Wave Mechanics
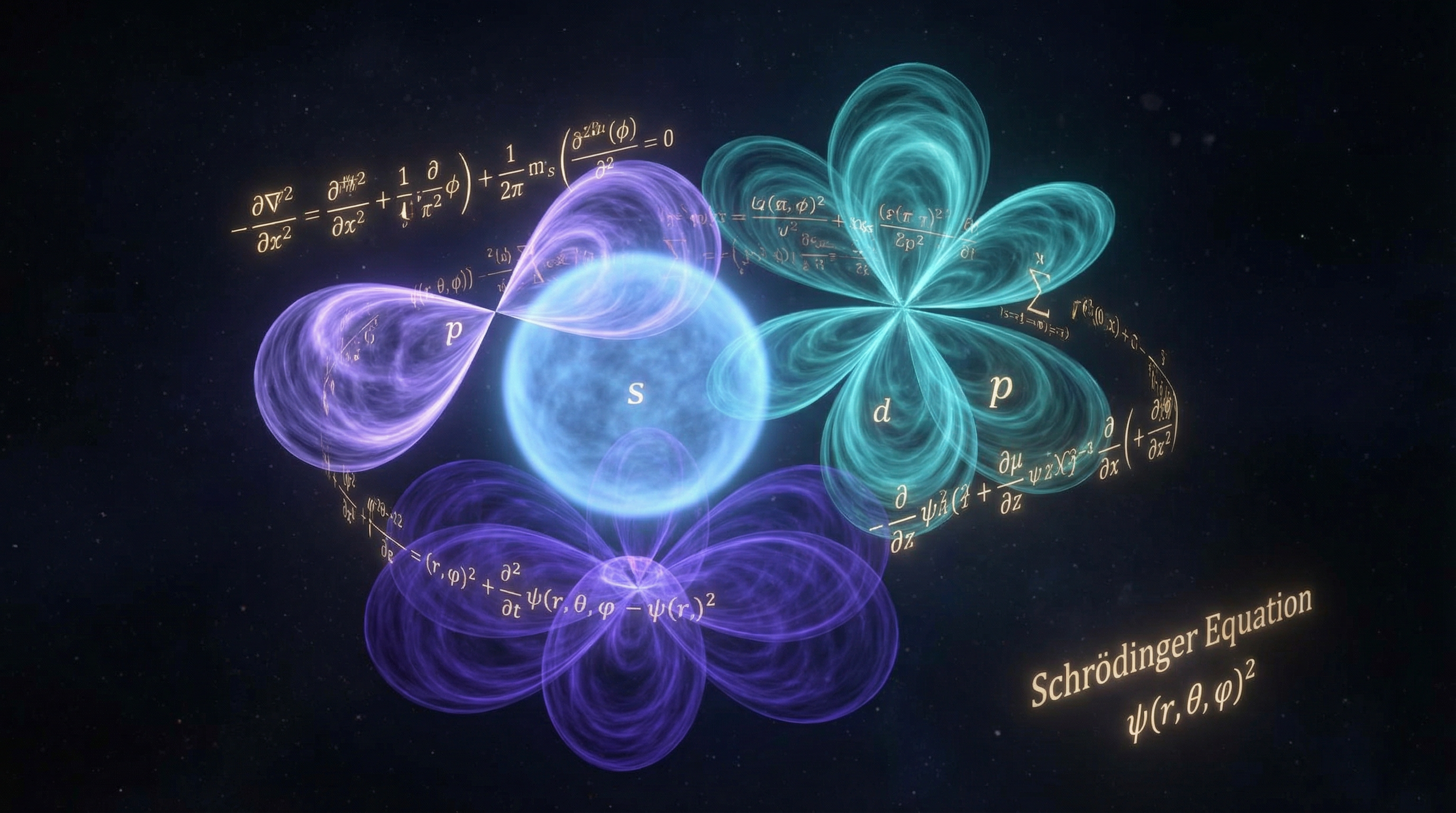
Schrödinger's Equation
Erwin Schrödinger, inspired by de Broglie's 1923 hypothesis (communicated to him by Victor Henri in 1925), developed a completely different formulation of quantum mechanics. During a Christmas 1925 holiday in the Alps, he derived the wave equation that bears his name.
In a series of four landmark papers published in 1926, Schrödinger solved the hydrogen atom exactly, developed perturbation theory, and treated the Stark effect. He also proved — much to his own and everyone's surprise — that wave mechanics and Heisenberg's matrix mechanics are mathematically equivalent.
1927
Era VIII — 1926–1927 Mathematical Formalization
Having two equivalent but superficially different formulations of quantum mechanics raised an urgent question: what is the mathematical structure of the theory? The answer came rapidly from several directions.
The Probabilistic Interpretation
Max Born provided the physical interpretation of Schrödinger's wave function $\psi(\mathbf{r})$. He proposed that $|\psi(\mathbf{r})|^2 d^3r$ is the probability of finding the particle in the volume element $d^3r$. This was a radical departure: the wave function does not describe a physical wave carrying energy — it encodes probabilities.
Dirac's Delta Function & Formalism
Dirac introduced the delta function $\delta(x-x_0)$ to represent a perfectly localized state — a mathematical tool that was later rigorously justified by Laurent Schwartz's theory of distributions. Dirac's bra–ket notation, developed over 1926–1930, provided an elegant unified language that encompassed both wave mechanics (continuous) and matrix mechanics (discrete).
Hilbert Spaces
David Hilbert, Lothar Nordheim, and later John von Neumann placed quantum mechanics on a rigorous mathematical foundation. The state of a quantum system is a vector in an abstract complex Hilbert space. Observables are self-adjoint operators. This reformulation — published by von Neumann in his 1932 masterwork Mathematische Grundlagen der Quantenmechanik — remains the standard mathematical framework today.
The Uncertainty Principle
Heisenberg derived the uncertainty relations from the commutation relation $[X,P]=i\hbar$: the product of the uncertainties in position and momentum cannot be smaller than $\hbar/2$. This is not a statement about measurement disturbance — it is a fundamental feature of quantum states. A quantum particle does not simultaneously possess a definite position and a definite momentum.
Density Operator & Measurement Theory
Von Neumann introduced the density operator $\hat{\rho}$ to describe mixed states — statistical ensembles of quantum states. He also developed the theory of quantum measurement, including the projection postulate: a measurement projects the state onto an eigenstate of the measured observable. This "collapse of the wave function" remains philosophically controversial to this day.
1997
Era IX — The Modern Era From QED to Bose–Einstein Condensation
The foundations laid in 1925–1927 unleashed a century of discovery. Quantum mechanics became the basis of atomic physics, nuclear physics, condensed matter physics, quantum optics, chemistry, and ultimately all of modern technology.
- 1927 Dirac quantizes the electromagnetic field — the first step toward quantum electrodynamics (QED), the quantum theory of light and matter.
- 1928 Dirac's relativistic equation — combines special relativity with quantum mechanics; predicts antiparticles (confirmed 1932 by Anderson's discovery of the positron).
- 1928 Felix Bloch — derives the band structure of electrons in crystals, founding solid-state physics and the theory of electrical conductivity.
- 1928 Gamow tunnel effect — quantum tunnelling explains radioactive alpha decay; a classically forbidden process becomes possible via the wave function.
- 1930 Rabi & NMR — Isidor Rabi develops nuclear magnetic resonance, the physical basis of modern MRI imaging.
- 1932 Anderson discovers the positron — confirms Dirac's prediction of antimatter; the first antiparticle ever observed.
- 1938 Kapitza discovers superfluidity in liquid helium-4 — a macroscopic quantum phenomenon where viscosity vanishes completely.
- 1946 Transistor invented (Bardeen, Brattain, Shockley at Bell Labs) — quantum mechanics of semiconductors; the foundation of all modern electronics.
- 1949 Quantum electrodynamics (QED) — Feynman, Schwinger, and Tomonaga develop the most precise theory in physics, predicting the electron's anomalous magnetic moment to 12 decimal places.
- 1957 BCS theory of superconductivity (Bardeen, Cooper, Schrieffer) — pairs of electrons form Cooper pairs and condense into a macroscopic quantum state of zero electrical resistance.
- 1962 Laser principle (Townes) and Josephson effect — quantum tunnelling of Cooper pairs through a thin insulating barrier; the basis of quantum voltage standards.
- 1977–1987 Bell inequality violations (Clauser, Aspect et al.) — experimental proof that quantum entanglement is real and local hidden-variable theories are ruled out.
- 1985 Scanning Tunnelling Microscope (Binnig & Rohrer, Nobel 1986) — images individual atoms using quantum tunnelling; the first direct visualization of atomic structure.
- 1995 Bose–Einstein Condensation (Cornell, Wieman, Ketterle) — a new state of matter predicted by Einstein in 1924: thousands of atoms occupy a single quantum ground state, forming a coherent matter wave. Nobel Prize 2001.
- 1997 Laser cooling & trapping — Nobel Prize to Steven Chu, Claude Cohen-Tannoudji, and William Phillips. Atoms cooled to microkelvin temperatures using photon recoil; enabling atomic clocks, BEC, and quantum simulation.

The Legacy
In less than three years — from Heisenberg's Helgoland paper in June 1925 to the formalization by von Neumann in 1927 — a small group of physicists in their twenties and thirties created a theory that overturned our deepest intuitions about reality. Quantum mechanics is today the foundation of chemistry, biology, materials science, electronics, and cosmology. Every smartphone, laser, MRI scanner, and GPS satellite is a working quantum device.